Clinical Presentation
Early CAS typically presents as single or multifocal ill-defined, enlarging, violaceous or dusky red macules or patches (Figure 1). Lesions often rapidly develop into raised nodules and plaques that may bleed and ulcerate. Other common symptoms include pain, edema, neuropathy, anemia, and weight loss; however, it is not uncommon for lesions to be asymptomatic.8,18-20 Nodular lesions are more common on the scalp, and patches are more common on the face and neck.16 Tumors typically extend into the dermis, and aggressive cancers may invade the subcutaneous tissue and fascia.2
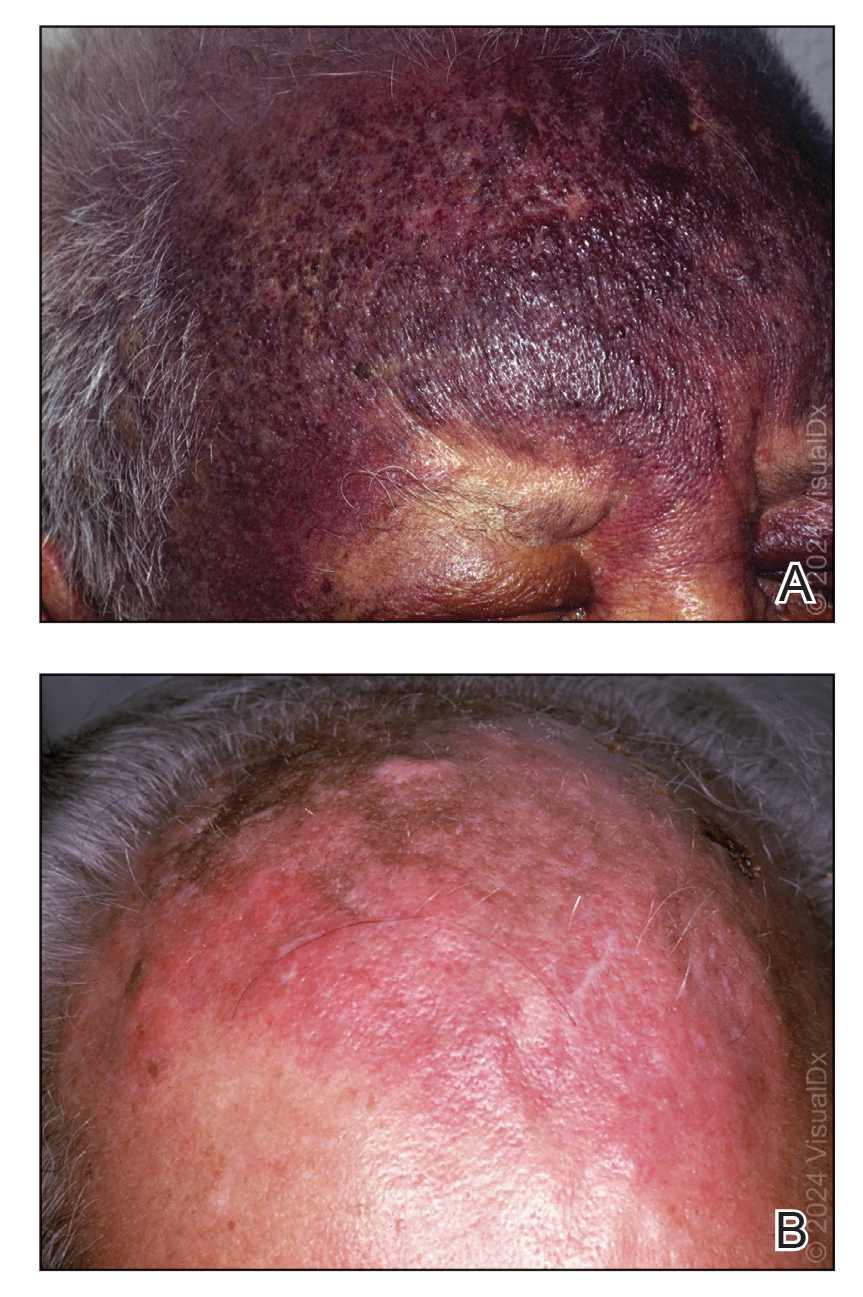
FIGURE 1. A, An extensive, deeply violaceous plaque with cobblestone appearance in areas on the forehead and a similar plaque on the left upper eyelid. B, An extensive reddish-brownish cutaneous angiosarcoma plaque on the scalp and forehead. Reprinted with permission from VisualDx (http://www.visualdx.com).
Cutaneous angiosarcoma may mimic ecchymosis, hemangioma, lymphangioma, edema, cellulitis, or scarring alopecia. Its nonspecific features make it difficult to recognize without dermoscopy or ultrasonography, which often results in delayed diagnosis and treatment. The median delay typically is 5 to 7 months and up to 1 year for some patients.7,16 Cutaneous angiosarcoma of the scalp tends to have a longer diagnostic delay than other areas of the body, which may be attributable to challenges in tumor identification and visualization by patients.16
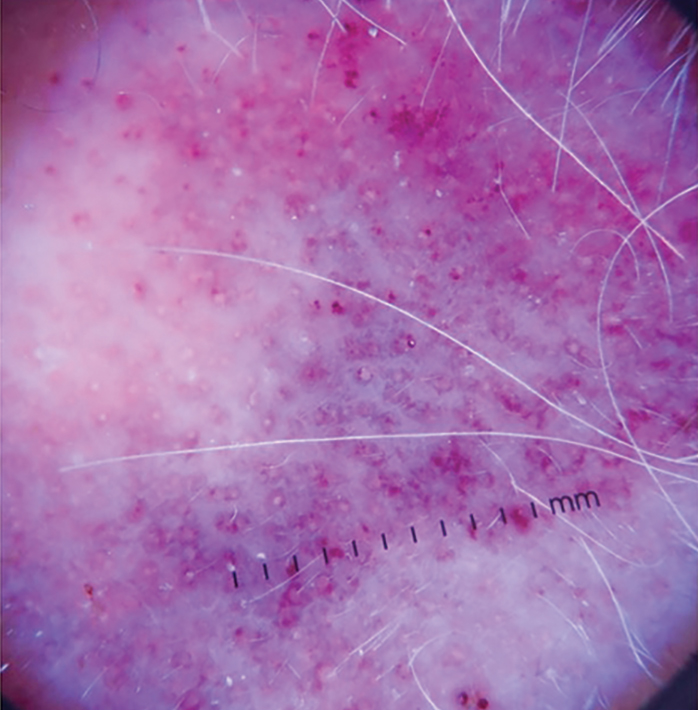
Dermoscopy and ultrasonography can aid in the diagnosis of CAS. Dermoscopy may demonstrate a range of colors with yellow, brown, or red areas in a violaceous background. Other reported features include white veils and lines, purple ovals, pink-purple “steamlike” areas, and atypical vessels (Figure 2).21-23 Dermoscopic findings may appear similar to other vascular tumors, such as hemangioma and Kaposi sarcoma, or nonvascular tumors, including amelanotic melanoma, Merkel cell carcinoma, and primary cutaneous B-cell lymphoma. Ultrasonography may show ill-defined, hypoechoic areas with anechoic reticular channels and a hypoechoic subepidermal layer.21 Other radiologic modalities, such as computed tomography, magnetic resonance imaging, or positron emission tomography, are nonspecific and are more useful in evaluating the extent of tumor spread in visceral angiosarcoma. Magnetic resonance imaging in CAS may indicate malignancy with the presence of high T2 and T1 signal intensity and high-flow serpentine vessels.24
Histopathology
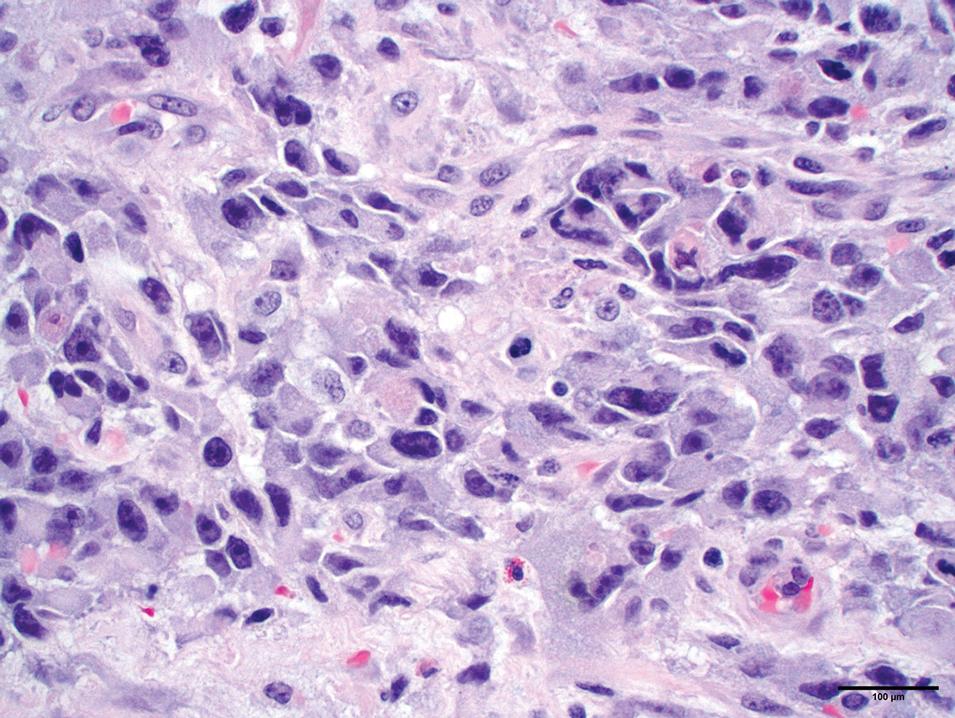
Histologically, angiosarcoma is characterized by anastomosing irregular vascular channels lined by a single layer of endothelial cells displaying slight to moderate atypia.25 These vascular channels dissect between collagen bundles and adipocytes. Monocyte infiltration may be observed.6 The neoplastic endothelial cells may present as spindle-shaped, round, polygonal, or epithelioid with eosinophilic cytoplasm. Histologic features differ based on the type of clinical lesion (Figure 3). In a study of CAS in Asian populations, nodular tumors showed solid sheets of pleomorphic spindle cells, many mitotic figures, and widely hemorrhagic spaces, whereas nonnodular tumors showed irregular vascular spaces dissecting collagen.16 Poorly differentiated tumors may present with hyperchromatic nuclei and prominent nucleoli, papillary endothelial formations, mitoses, and possible hemorrhage or necrosis.2,6,8 Histologic specimens also may reveal calcified bodies and hemosiderin particles.19 Angiosarcomas typically are invasive without a clear capsule or border.6
Secondary CAS in the setting of lymphedema and radiation therapy has MYC amplification and is positive for MYC via immunohistochemistry, which is uncommon in primary angiosarcoma.26 Immunohistochemical staining of tumor specimens is helpful to confirm the diagnosis of CAS. These markers include CD31, CD34, CD117, cytokeratin, vimentin, epithelial membrane antigen, factor VIII–related antigen, Ulex europaeus agglutinin-1, von Willebrand factor, and VEGF.6,19,27,28 Notably, advanced angiosarcomas with progressive dedifferentiation often lose these markers.
Treatment
Surgery—The majority of patients treated for CAS undergo surgical resection, as surgery has been shown to have the best prognosis for patients.5,9,10,13,15 Achieving R0 resection (microscopically negative margins) is the most important factor in determining the success of treatment, with incomplete surgical resection resulting in higher rates of systemic and local spread.29 Abraham et al8 found that the median disease-specific survival of patients with microscopically negative margins was 83.7 months; patients with microscopically positive and grossly positive margins had median disease-specific survival of 63.4 and 18.1 months, respectively. In a case series of patients undergoing resection with negative surgical margins, 4 patients demonstrated no evidence of local recurrence or systemic disease at an average of 4.3 years after therapy, and the other 4 patients each had 1 local recurrence but were disease free an average of 4.8 years after removal of the recurrent lesion. In a series of 27 patients with positive surgical margins, there was local recurrence within 2 years for most patients.12
Large tumors invading nearby structures may not be amenable to surgical resection because of extensive local growth, propensity for skip lesions, and localization near vital organs of the head and neck.5,7 The extended delay in diagnosis often seen in CAS allows for advanced local progression, resulting in large areas of resection. In a case series (N=8), the average surgical defect measured 14.3×11.8 cm, necessitating reconstruction with either a tissue flap or split-thickness skin graft in every case because primary closure was not possible. More than 80% of patients in this study still had positive margins after surgery, necessitating the use of additional chemotherapy or radiation to eradicate remaining disease.7 In several studies, multimodality therapy was associated with improved overall survival.7,14,30